Molar concentration
If we know the molar concentration of our solution and the formula of the solute, then we can determine the amount of solvent present in the solution, both in moles and in grams.

We have to make sure that before we add the atomic masses together, we multiply each of the atomic masses for a specific atom by the number of atoms of this type present in the molecule. In particular, to find how many moles of the solution we have, we can look up the atomic mass of each atom present in the molecule in the periodic table, and then divide the total mass of the substance by the total atomic weight of atoms in the molecule. To determine the amount of the substance we could use the molecular formula for this substance and information about the mass of this substance that is present in the solution. To find molar concentration we need to know the amount of substance and the total volume of the solution. It can also be measured in moles per another unit of volume. Molar concentration is measured in moles per unit of volume, for example in moles per liter or moles per cubic meter. As a result, all or parts of the solvent stop being dissolved in the solution, and the concentration is decreased. Sometimes raising or lowering the temperature changes the solubility. In this case, the concentration of the solution will increase. It is also possible for the solvent to evaporate while the amount of the solute remains the same, as the temperature increases. Temperature can cause some solvents to expand, and if the solute does not expand with the solvent, then the molar concentration decreases. Molar concentration can be affected by temperature, although this depends on the substances present in this solution. It can be found using the periodic table. Weight of one mole of various substances. This is the case if our solvent is water, and if the amount of solute is small enough that its mass and volume are insignificant - but this is not always the case.
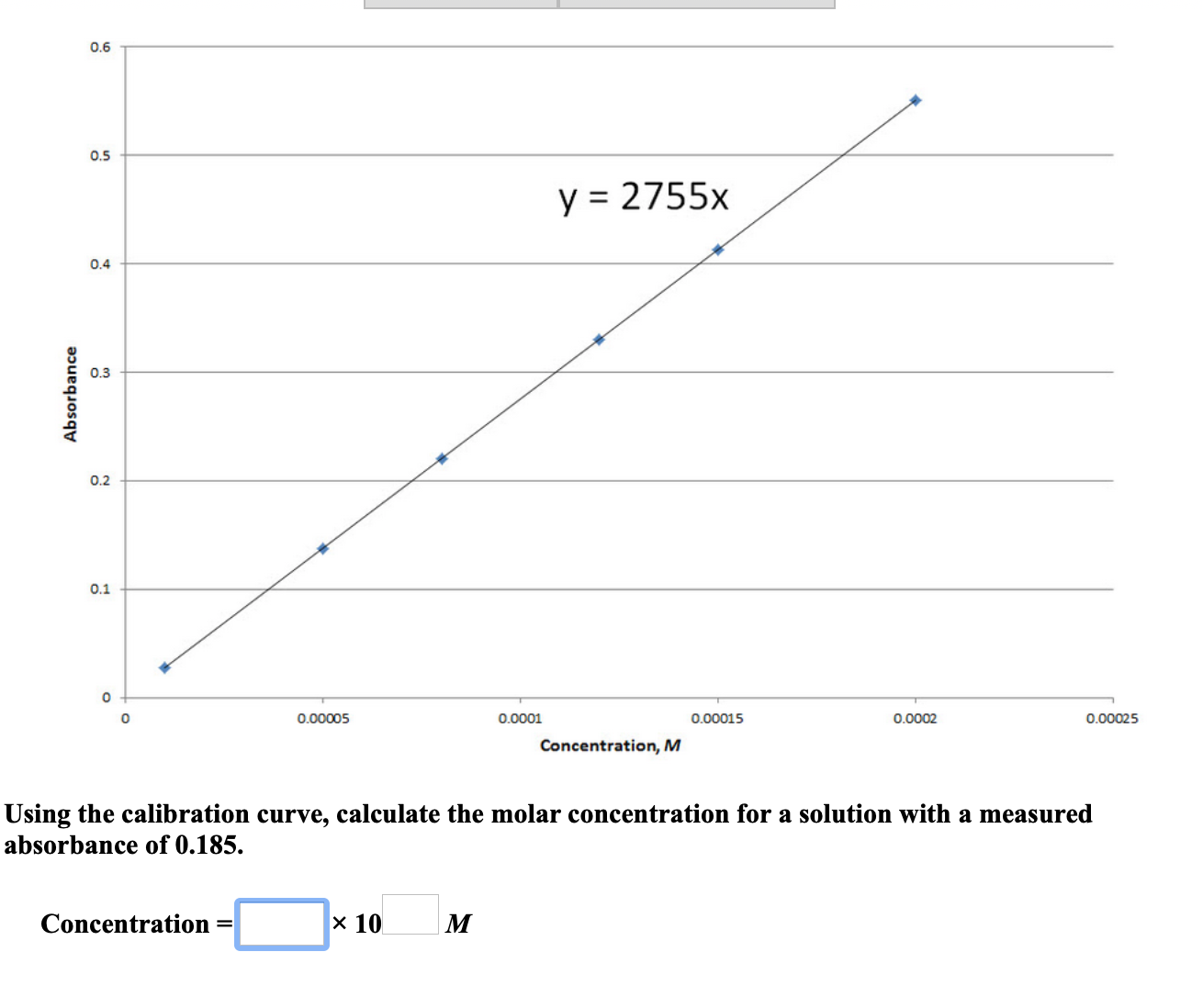
In some cases, values for molarity and molality of a solution are very close. Unlike molarity, molality is the ratio of the amount of substance of the solute to the mass of the solvent, and not to the mass of the entire solution. We have to be careful not to confuse molarity with another related property, molality. Molar concentration is also sometimes called molarity. We can also use moles to measure other particles such as molecules or electrons, but we would need to specify which particles are in use in this case.

The elementary particles most commonly used when working with moles are atoms. Otherwise, we would need to use either very large numbers or very small quantities (for mass or weight) that are hard to work with and impossible to measure using currently available measuring devices. Moles are very convenient to use for substances that are in quantities small enough, that can be easily measured using household or industrial measuring devices. One mole is defined as the number of atoms in 12 grams of carbon-12, which is about 6×10²³ atoms. Because there are vast numbers of elementary entities even in a small volume of a substance, we use special units called moles for the amount of substance. Here the amount of substance is measured as the number of elementary entities (e.g. The substance in our case is the solute, while the volume is measured for the entire solution, even if it has other solutes in it. Here we consider molar concentration, which is measured as the ratio of the amount of substance in moles to the total volume of the solution. One mole of carbon is the amount of the substance that has the same number of atoms as in 12 grams of carbon-12, or more specifically 6×10²³ atoms.Ī concentration of a solution can be measured in different ways, for example by measuring the ratio between the mass of the solute and the total volume of the solution.
